|
3/22/2023 0 Comments Hydrogen atomic orbitals 
Both formats are vector graphics, consisting of curved and straight lines and text such file types can be enlarged without pixelation, and SVG images can be edited (for example, with Inkscape or Gimp). (3-5, 11, 12, 17, 23)) Images in scalable vector graphics (SVG) and enhanced metafile (EMF) formats are provided in the Supporting Information. These “polar plots” distort the relationship between position and the value of ψ. (Previous implementation of isosurfaces in gnuplot (19) and Winplot, (24) a similar program, was limited to the angular wave function. The parametric equations were used to generate image files for the isosurfaces using gnuplot. In all, the parametric equations represent 52 orbitals. The derivations and complete details for the parametric equations are given in the Supporting Information. (For example, the d xy orbital constitutes 45° rotation around the z-axis.) The other orbitals are generated through rotations of the d x 2– y 2 orbitals around axes. The d x 2– y 2 orbitals have a similar form as an ellipse in plane geometry for x and z around the y-axis and for y and z around the x-axis at a specific value of r. The d x 2– y 2, d xy, d yz, and d xz orbitals for 4 ≤ n ≤ 6 were generated with new parametric equations. (The p x and p y orbitals can be generated by exchanging the variables.) In this work, the surface is generated using the cylindrical symmetry of these orbitals around the z-axis in addition, expressions for the additional orbitals are derived. (10) He described contour line parametrizations for the 2p z, 3p z, and 3d z 2 orbitals in the xz-plane. and 6d z 2 orbitals were generated by using and extending the parametric equations for contour lines derived by Scaife. More details about the Lambert W function and its implementation are found in the Supporting Information.) These are the first parametric equations for f and g orbitals in the literature, although they cannot be extended to higher principal quantum numbers. (26, 27) The wave functions for these orbitals can be manipulated such that the Lambert W function can be used. (The Lambert W function is the inverse of the function f( x) = x e x. These parametric equations use the Lambert W function. The distance from the origin r was related to the angles θ (the polar angle from the z-axis) and φ (the azimuthal angle around the z-axis) in spherical coordinates. The 2s, 2p, 3d, 4f, and 5g orbitals were generated with a new set of parametric equations. The orbitals were generated using three sets of parametric equations: 1. These representations are generated with gnuplot, a readily available, free, open-source program. Here we generate vector graphic images for a large set of orbitals using parametric equations, many of which are newly described. 
(8, 12, 13, 21)) The use of software has allowed for orbital plotting for pedagogical purposes, with varying levels of complexity and success. (The marching cubes algorithm is a common algorithm for isosurfaces, (25) and other algorithms have been used for computerized plotting of orbitals. The alternative to these equations is to generate the isosurfaces or contour lines implicitly through an algorithm that may require programming or software with the ability to implement them. (1, 2) To date, no parametric equations have been derived for the 4f or 5g orbitals or any orbitals with n > 3. (18) Before that, Scaife introduced parametric equations for contour lines in the xy-, yz-, and yz-planes for orbitals with n ≤ 3, (10) which were an expansion from earlier approaches. Ramachandran and Kong derived parametric equations to generate atomic orbital isosurfaces for 2s, 2p, 3p, 3d x 2– y 2, and 3d z 2 orbitals in terms of r, the distance from the origin. The easiest method to generate an isosurface is to use an equation for the surface. The values of ψ and ψ*ψ, the probability density, are closely related. 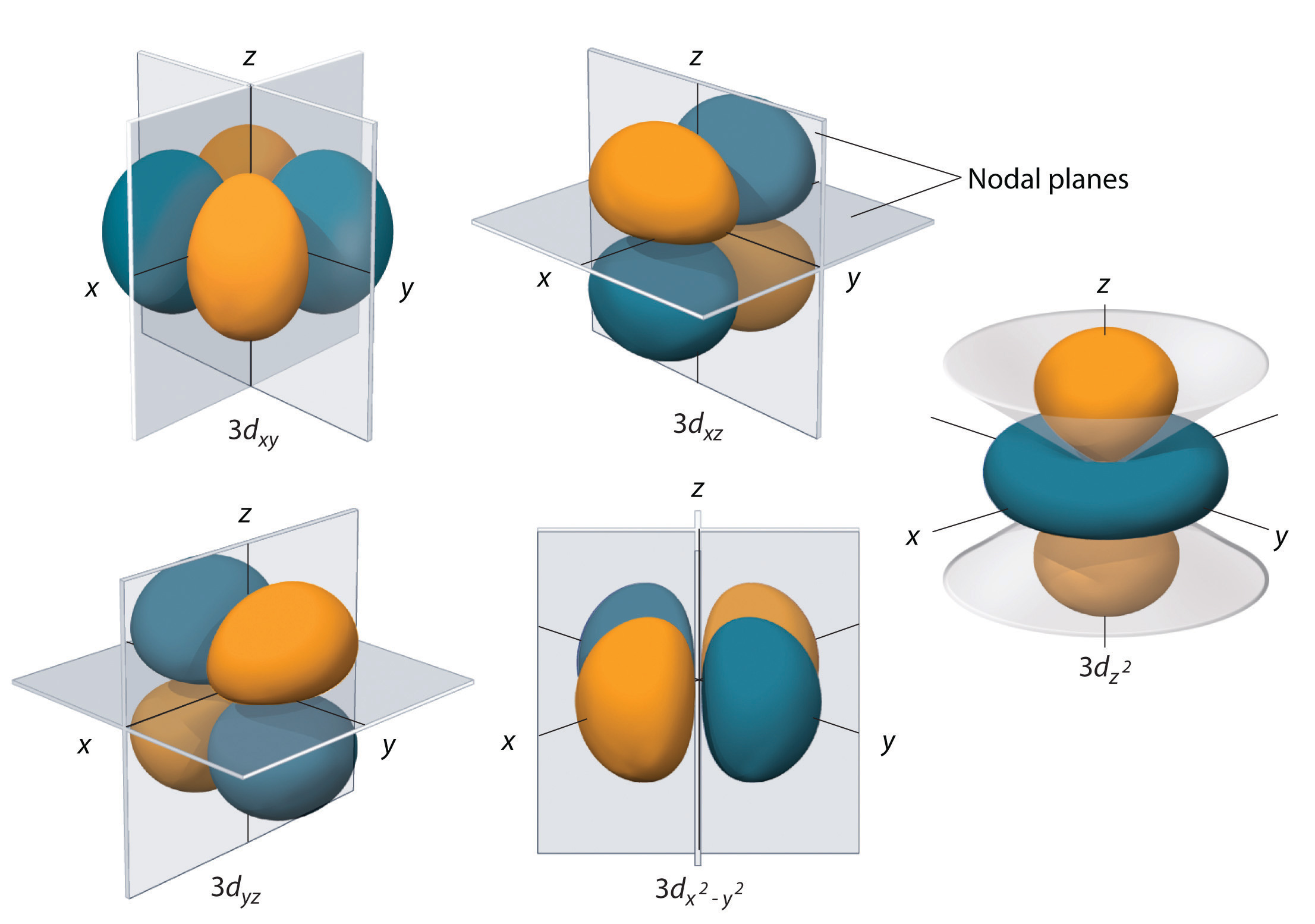
(1-24) In these representations, a surface of a fixed value of the wave function ψ is plotted in analogy to a contour map. 
Contour plots as isosurfaces or contour lines in a plane are the most familiar representations of the hydrogen wave functions. Atomic orbitals are a theme throughout the undergraduate chemistry curriculum, and visualizing them has been a theme in this journal. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
